2,130
2,13
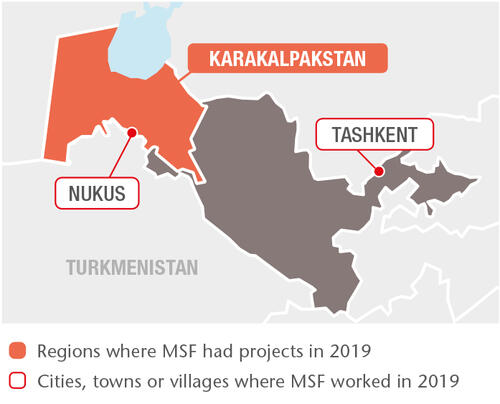
In January 2019, we started to enrol new patients in Tashkent region for our multi-site TB PRACTECAL clinical trial. This trial was launched in 2017 in Nukus, Karakalpakstan, to evaluate the safety and effectiveness of two of the newer TB drugs – bedaquiline and pretomanid – on a much shorter and more tolerable regimen lasting just six months. By the end of 2019, we had 166 patients enrolled across the two sites.
We also work with the Ministry of Health at our project in Tashkent, which focuses on improving the availability of integrated care for HIV patients co-infected with TB, sexually transmitted infections and hepatitis C through a ‘one-stop shop’ model. Since the end of 2018, we have been providing services to high-risk groups, such as sex workers and people who inject drugs, in the greater Tashkent area. We are also moving forward with plans to use the TB LAM test to facilitate diagnosis of TB in HIV patients.
The Karakalpakstan TB project implements a comprehensive patient-centred model of care. In 2019, 2,130 patients received treatment as part of this programme, 660 of whom were treated for drug-resistant TB (DR-TB) and 1,470 for drug-sensitive TB. Towards the end of the year, as part of our commitment to introduce best practice guidance, we supported the Ministry of Health in the rollout of TB care protocol based of the revised WHO consolidated guidelines for DR-TB treatment in Karakalpakstan. In line with the policy recommendations of the WHO consolidated guidelines, we upgraded the laboratory capacity by introducing drug-susceptibility testing for both new and repurposed TB drugs (bedaquiline, linezolid, clofazimine, moxifloxacin, amikacin and pretomanid).