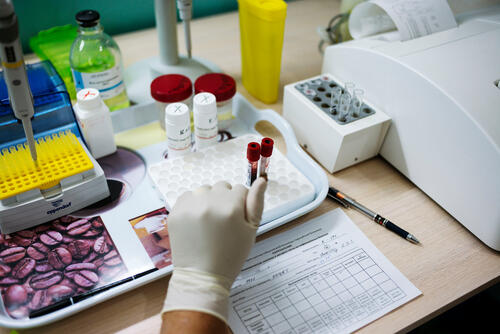
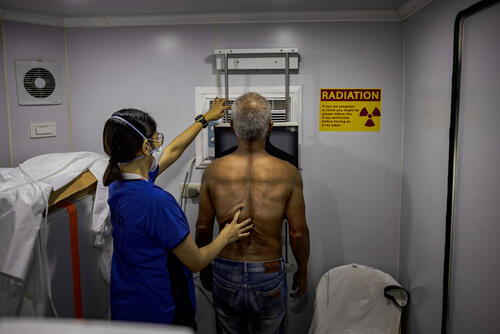
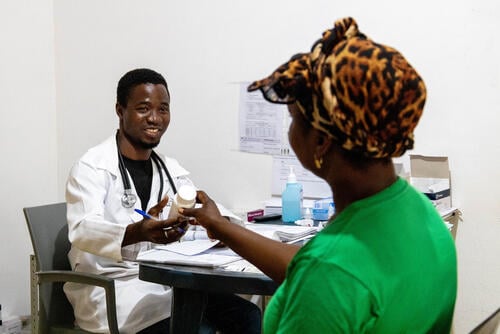
Obsolete treatments, the lack of an effective vaccine, and the lack of suitable diagnostic tools make it difficult to control the global TB epidemic. In 2022, 7.5 million people across 192 countries and areas were diagnosed with TB.
Some gains have been made in recent years; the first new TB drugs in half a century and the trial of a shorter course of treatment for drug-resistant TB. But the harsh reality remains - 10.6 million people fell sick with TB and 410,000 developed multidrug-resistant TB (MDR-TB) in 2022, which is much harder to treat. Only about one-third of people with DR-TB accessed treatment in 2021. The majority go undiagnosed and therefore untreated.

22,700
22,7
2,700
2,7
Quick facts about tuberculosis


The most widely-used test for diagnosing active TB in low- and middle-income countries relies on examining a patient’s phlegm under a microscope. This method, developed nearly 140 years ago, detects less than half of all TB cases and largely fails to detect the disease in children and people co-infected with HIV – who usually can’t produce the sputum needed – and those with drug-resistant forms of TB. A diagnostic test, Xpert MTB/RIF, was introduced and we use it in many of our projects, but it can't be used in many resource-limited settings.


A course of treatment for uncomplicated TB takes a minimum of six months. Drug-resistant TB (DR-TB) treatment requires taking a cocktail of drugs and can take up to two years or more, but trials are ongoing to treat it in six to nine months. It is vital that a patient completes their entire course of treatment, even when they start to feel better; incomplete treatment can lead to drug resistance developing. Decentralising treatment by having people cared for at home can help them adhere to treatment and overcome any obstacles they may face.


When patients are resistant to the two most powerful first-line antibiotics (rifampicin and isoniazid), they are considered to have multidrug-resistant TB (MDR-TB). Treatment is long and arduous, with the drugs having many potential side-effects, including psychosis and deafness. People can take as many as 20 pills a day for two years - only to have a cure rate of little better than half. Extensively drug-resistant TB (XDR-TB) occurs when a patient is resistant to second-line drugs. Only a third of people with XDR-TB are cured.

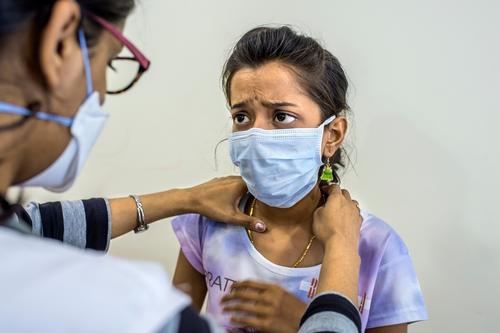
Most drugs to treat TB are old and toxic. But in the last decade, bedaquiline, delamanid and pretomanid, three new TB drugs - the first in half a century - were developed, giving patients hope and treatment providers options. But making these drugs available to patients who desperately need them has been painfully slow. We estimated that, in 2016, only around one in eight people with DR-TB who could have benefited from the lifesaving newer drugs bedaquiline and delamanid were treated with them.


Most of the drugs used to treat TB are decades old, while some were not originally developed for TB use, and/or have toxic side effects. Treatment itself for DR-TB can take up to two years. For these reasons, research into new, shorter, more effective drug regimens is urgently needed. We are participating in two clinical trials, EndTB and PRACTECAL, to find new treatment regimens. The EndTB trial enrolled its final patients in October 2021. In the same month, the PRACTECAL trial announced in preliminary results that its six-month, all-oral regimen cured nine out of 10 patients.

Countries and donors must sustain investment in fight against tuberculosis in children

MSF hands over drug-resistant tuberculosis project in Mumbai after 18 years

TB treatment decision algorithms: promising early data highlight their added value in diagnosing children

Urgent action needed as governments and donors fail children with tuberculosis

Cross-cutting lessons: results from the TB ALGO PED study

Action needed on HIV and tuberculosis amid South Sudan’s crises

Bringing healthcare to artisanal miners in Zimbabwe

A timeline of tuberculosis treatment success in Tajikistan

Year in Review 2023
MSF Tuberculosis Research
fieldresearch.msf.orgWe produce important research based on our field experience. So far, we have published articles in over 100 peer-reviewed journals. These articles have often changed clinical practice and have been used for humanitarian advocacy. Read all our Tuberculosis-related articles on our dedicated Field Research website.