Discover all our latest news, stories and publications. Use the filter to get to the content you're looking for.

War-torn minds: navigating mental health issues amid war in Ukraine

Malaria on the rise in Madagascar as climate change leaves healthcare out of reach

Charges in case against rescues at sea dropped in Italy
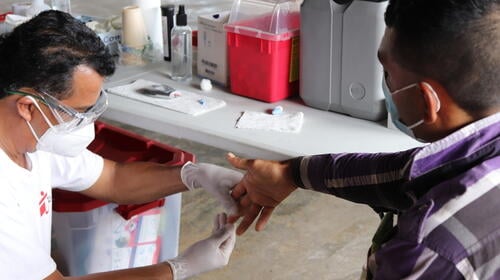
MSF highlights urgent need to care for patients with chronic kidney disease

MSF office and pharmacy in Rakhine state destroyed in fire amid ongoing violence

MSF concerned over deportations of Afghans from Pakistan

Five things to know about the humanitarian crisis in northern Rakhine

After a year of war in Sudan, a rapid scale up of response is needed to avoid catastrophe

In Port-au-Prince, the “violence is like gangrene, spreading and threatening us all.”

In Sudan, “the needs are growing by the day, but the response is deeply inadequate”

MSF condemns missile attack which destroys office, injures staff in Donetsk

Why we won't accept the narrative of ‘regrettable incidents’ in Gaza

Surviving sexual violence in the camps of Benue

How the Israeli army besieged and attacked Nasser hospital

Eight years in Kirkuk: following along the journey of Hawija’s recovery

Urgent vaccination campaign needed to halt deadly measles outbreak in Western Equatoria state

Displaced by war, people in eastern Sudan urgently need food, water and shelter
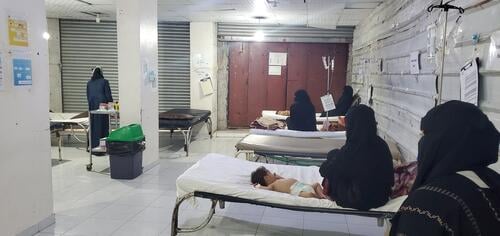
The upsurge in measles cases shows no sign of abating

MSF rescue ship detained in Italy following threats by Libyan Coast Guard

Against stigma and drug-resistance: five years of treating tuberculosis in Ukraine

Five months into border conflict, displaced people’s needs are on the rise

Five things to remember a decade after the Ebola outbreak in West Africa

EU-sponsored shameful abuse in the Central Mediterranean must end

Numbers of malnourished women on the rise in Yemen

How we deliver medical humanitarian assistance
Everywhere we work, the circumstances are unique. Nonetheless, our programmes generally follow a common set of practices designed to make sure our resources and expertise are used to maximum effect.




