The disease is easy enough to treat, but access to the most effective treatments remains inadequate. Over 95 per cent of all malaria deaths occur on the African continent. Impregnated mosquito nets are expensive and out of reach for many. The parasite which causes malaria is beginning to show resistance in parts of Asia to the most effective drug we have. And there are no new drugs in the development pipeline, meaning we could be left without effective options in the future.
Quick facts about malaria


Mosquitoes breed in water and they are especially plentiful during the rainy season. If the most basic prevention is to avoid bites by sleeping under insecticide-treated bed nets, the natural breeding ground of the insect needs to be eliminated. Spraying insecticide and drying sources of stagnant water are strategies used for controlling the proliferation of mosquitoes.


Diagnosing malaria is done by using either a rapid diagnostic test or a microscope with a skilled laboratory person. A rapid diagnostic test uses blood from a finger prick and is relatively cheap and easy to perform—community health workers with no previous training can quickly learn to do them. In some areas neither test is available, so health workers must diagnose sick children based on symptoms alone - leading to over-diagnosis of malaria, while the real cause of the symptoms is left untreated.


The most effective treatment for malaria is a two-drug combination called artemisinin-based combination therapy (ACT). These drugs cure most infections in three days. Early treatment is essential to avoid severe malaria, which requires hospitalisation with intravenous antimalarials. Patients with severe malaria often need blood transfusions, requiring a safe, adequately-stocked blood bank—difficult to achieve in under-resourced areas. Without treatment, or with significantly delayed treatment, severe malaria kills.


The WHO recommendation of ACT, which contains the drug artemisinin, in 2001 was a major breakthrough, replacing old, ineffective treatments such as chloroquine, or quinine. But the malaria parasite has developed resistance to artemisinin in parts of Southeast Asia, meaning that ACT may no longer be effective. If the spread of resistance cannot be stopped, it represents a major risk to public health, and would most likely lead to a resurgence of malaria, since there aren’t currently any new drugs available.

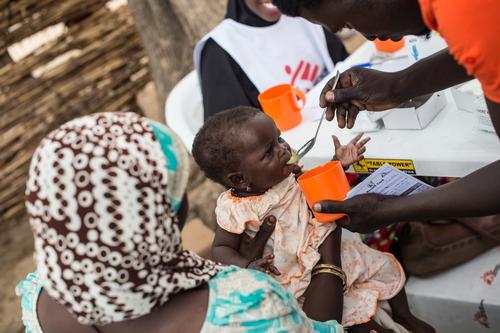
In Africa’s Sahel region, malaria is highly seasonal and most cases happen during a 4-5 month period. In 2012, MSF piloted a seasonal malaria chemoprevention (SMC) in Mali, Niger and Chad, now used in several countries. SMC involves providing antimalarial drugs to prevent infection among those most at-risk - children under five. Malaria can also be prevented by avoiding being bitten by the mosquitoes that carry the parasite, through the use of insecticide-treated mosquito nets, unavailable for many.

Malaria on the rise in Madagascar as climate change leaves healthcare out of reach

MSF launches mass vaccination campaign amid deadly hepatitis E outbreak in South Sudan
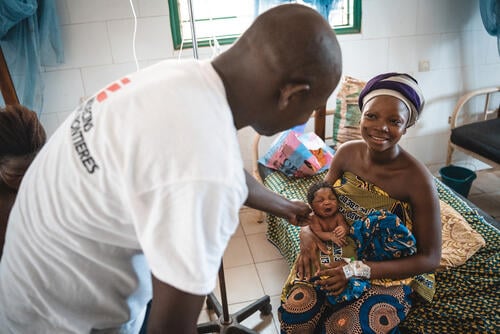
Limiting maternal and child loss in communities in Benin

Aid woefully inadequate for returnees stuck in deplorable conditions in Renk

Two decades of combatting the world’s most neglected diseases

Three things to know about the humanitarian situation in Niger
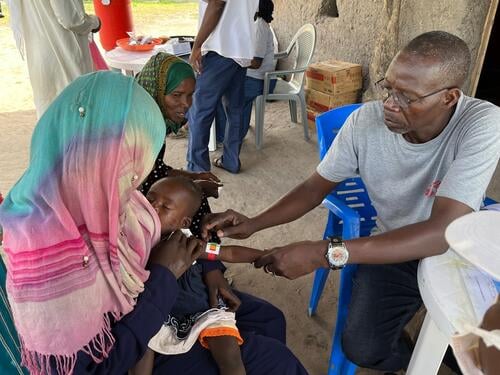
Community-focused projects aim to reduce rising malnutrition in Chad

Health emergency in Central African Republic requires urgent response

Communities in South Sudan prepare for flooding ahead of rainy season
Research & Analysis
Swapping production of malaria tests for covid-19 tests requires an urgent response
Plasmodium prevalence and artemisinin-resistant falciparum malaria in Preah Vihear Province, Cambodia: a cross-sectional population-based study
Population pharmacokinetics of quinine in pregnant women with uncomplicated Plasmodium falciparum malaria in Uganda

Malaria control in emergencies: time for action
Can Timely Vector Control Interventions Triggered by Atypical Environmental Conditions Prevent Malaria Epidemics? A Case-Study from Wajir County, Kenya

Making the Switch: Ensuring access to improved treatment for severe malaria in Africa

Malaria: Introducing ACT from Asia to Africa
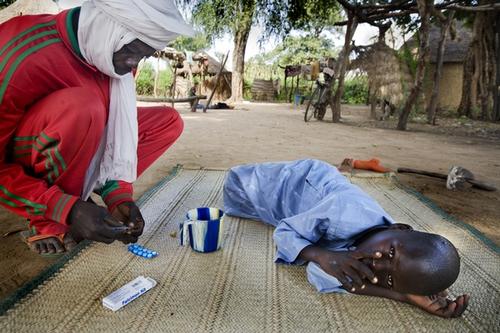
Full Prescription: Better malaria treatment for more people, MSF's experience
MSF Malaria Research
fieldresearch.msf.orgWe produce important research based on our field experience. So far, we have published articles in over 100 peer-reviewed journals. These articles have often changed clinical practice and have been used for humanitarian advocacy. Read all our Malaria-related articles on our dedicated Field Research website.





